UC Berkeley Press Release
Whales evolved biosonar to chase squid into the deep
BERKELEY – Behind the sailor's lore of fearsome battles between sperm whale and giant squid lies a deep question of evolution: How did these leviathans develop the underwater sonar needed to chase and catch squid in the inky depths?
Now, two evolutionary biologists at the University of California, Berkeley, claim that, just as bats developed sonar to chase flying insects through the darkness, dolphins and other toothed whales also developed sonar to chase schools of squid swimming at night at the surface.
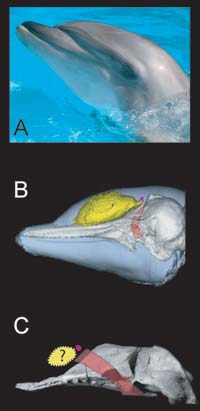 A fatty melon (yellow) cradled in a concave area of the skull (B) allows the bottlenose dolphin to focus clicks it makes with muscles (purple) below its nostril. Echoes help the dolphin locate food such as squid. The skull of Simocetus (C), an extinct ancestor of today's toothed whales, shows a slight concavity that may have held a similar melon. Analogous to today's dolphins, the extinct whale may have used its large nostril muscles (purple) to amplify the clicks powered by their nasal passage (red). (CT scan courtesy of T. W. Cranford & M. F. McKenna/San Diego State University. Simocetus courtesy of Nick Pyenson/UC Berkeley. Bottlenose dolphin courtesy of Wikipedia.) |
Because squid migrate to deeper, darker waters during the day, however, toothed whales eventually perfected an exquisite echolocation system that allows them to follow the squid down to that "refrigerator in the deep, where food is available day or night, 24/7," said evolutionary biologist David Lindberg, UC Berkeley professor of integrative biology and coauthor of a new paper on the evolution of echolocation in toothed whales published online July 23 in advance of its publication in the European journal Lethaia.
"When the early toothed whales began to cross the open ocean, they found this incredibly rich source of food surfacing around them every night, bumping into them," said Lindberg, former director and now a curator in UC Berkeley's Museum of Paleontology. "This set the stage for the evolution of the more sophisticated biosonar system that their descendents use today to hunt squids at depth."
Lindberg and coauthor Nick Pyenson, a graduate student in the UC Berkeley Department of Integrative Biology and at the Museum of Paleontology, reconstructed this scenario after looking at both whale evolution and the evolution of cephalopods like squid and nautiloids - relatives of today's chambered nautilus - and relating this to the biology of living whales and cephalopods.
All toothed whales, or odontocetes, echolocate. The baleen whales, which sieve krill from the ocean and have no teeth, do not. The largest of the toothed whales, the sperm whale, grows up to 60 feet long and dives to 3,000 meters - nearly two miles - in search of squid. Though poorly known because they live entirely in the deep ocean, the many species of the beaked whale dive nearly as deep. Belugas and narwhals descend beyond 1,000 meters, while members of the dolphin family - porpoises, killer whales and pilot whales, for example - all can dive below the 200-meter mark where sunlight is reduced to darkness.
According to Pyenson, who focuses on the evolution of whales, the first whales entered the ocean from land about 45 million years ago, and apparently did not echolocate. Their fossil skeletons do not have the scooped forehead of today's echolocating whales, which cups a fatty melon-shaped ball that is thought to act as a lens to focus clicking noises.
Skulls with the first hints of a concave forehead and potential sound-generating bone structures arose about 32 million years ago, Pyenson said, by which time whales presumably had spread throughout the oceans. Whales had developed underwater hearing by about 40 million years ago.
According to Lindberg, whale biologists had various theories about echolocation, including that whales developed this biosonar soon after entering the water as a way to find food in turbid rivers and estuaries. The evolution of toothed whales, however, indicates otherwise. Whales first occupied the ocean, and only later invaded rivers. Other experts have proposed that development of echolocation coincided with global cooling around 33.5 million years ago, though a mechanism was not specified.
The most convincing explanation, that echolocation allowed whales to more efficiently find food in the darkness of the deep ocean, ignores the question of evolution.
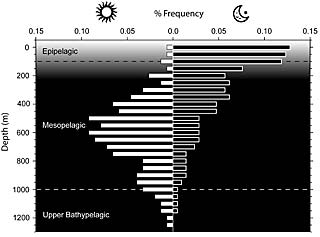 Cephalopod species diversity at depth during the day (left panel) and night (right panel), showing how many cephalopods descend into darkness during the day and return to surface waters at night. UC Berkeley evolutionary biologists argue that whales feeding at night on squid and nautilus at the surface developed echolocation to follow them into the black depths during the day. (Data from Roper & Young [Smithson. Contr. Zool. 209: 1975], Nesis & Nikitina [Russ. J. Aquat. Ecol. 4: 1995] and Moiseev [Bull. Mar. Sci. 49: 1991]) |
"How did the whales know there was a large supply of food down in the dark?" asked Lindberg, noting that cephalopods are the most abundant and high-energy resource in the ocean, eaten by 90 percent of all toothed whales. "What were the intermediate evolutionary steps that got whales down there?"
Lindberg, a specialist in the evolution of marine mollusks, noted that cephalopods have migrated up and down on a daily "diel" cycle for at least 150 million years. At the time whales developed biosonar, nautiloids dominated the oceans. Lindberg and Pyenson propose that whales first found it possible to track these hard-shelled creatures in surface waters at night by bouncing sounds off of them, an advantage over whales that relied only on moonlight or starlight. This would have enabled whales to follow the cephalopods as they migrated downwards into the darkness during the day. Today, the largest number of squid hang out during the day at about 500 meters below the surface, though some go twice as deep. During the night, however, nearly half the squid are within 150 meters of the surface.
Over the millennia, cephalopod species in general - and especially shelled cephalopod species - fell as the number of whale species boomed, possibly because of predation by whales. Then, about 10 million years ago, the whales seem to have driven the nautiloids out of the open ocean into protected reefs. Lindberg said that the decline in nautiloid diversity would have forced whales to perfect their sonar to hunt soft-bodied, migrating squid, such as the Teuthida, which in the open ocean are typically two feet long or bigger and range up to the 40-foot-long giant squid.
"Whales didn't need to have a very sophisticated sonar system to follow the nautiloids, they could just home in on the hard part," Lindberg said. Only later , he added, did they "develop a complex system with finer resolution to detect and capture soft-bodied squid."
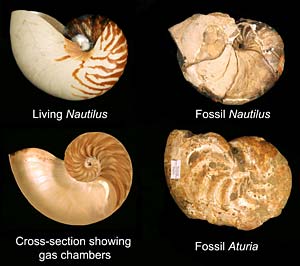 Comparison of the living chambered nautilus (left) and fossil nautiloids (right). (David Lindberg/UC Berkeley) |
"Whales, like bats, developed a sensory system for seeing with sound, and every single toothed whale echolocates in a different way, just like how different bat species echolocate in different ways," Pyenson said. Whales also partition the water column, specializing in harvesting squid at specific depths, just as bats partition the tree canopy and preferentially hunt insects at specific heights.
Lindberg noted that whales and bats are strong examples of convergent evolution to take advantage of unexploited food resources: nocturnal insects, in the case of non-migrating insectivorous bats, and nocturnal cephalopods, in the case of whales. And just as predominately migrating fruit bats do not echolocate, so filter-feeding baleen whales that depend on dense seasonal resources lack biosonar.
Lindberg and Pyenson used existing data on whales and cephalopods to reach their conclusions, drawing upon aspects of tectonics, paleontology, physiology, ecology, anatomy and biophysics. In the same way, "thinking from an evolutionary perspective about existing data from biology, paleontology and ecology could answer questions about the origin of echolocation in bats, shrews and other animals," Lindberg said.
The work was supported in part by the Remington Kellogg Fund and the Doris O. and Samuel P. Welles Research Fund of the UC Berkeley Museum of Paleontology and by a graduate research fellowship from the National Science Foundation.

